Racing Against the Pandemic
Tuesday, March 24, 2020
arsip tempo : 173075270340.
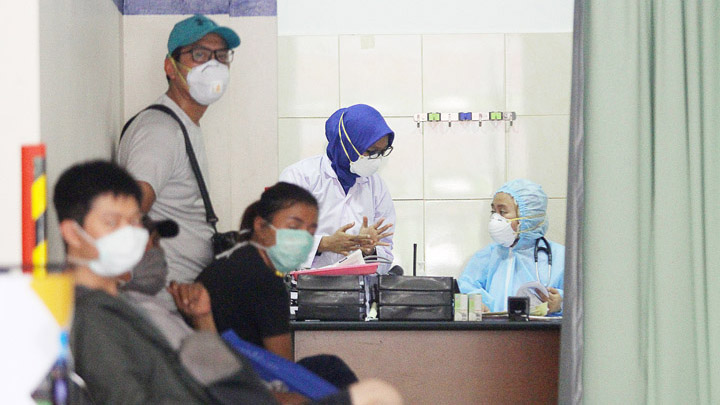
PRESIDENT Joko Widodo needed 17 days to decide on using the rapid test kit in order to detect early those infected with the novel coronavirus. Since President Jokowi announced the first two confirmed Covid-19 cases in Indonesia on March 2, the disease has rapidly spread, infecting over 500 individuals in 20 provinces and causing 48 deaths. “I want to increase the number of rapid test kits,” said Jokowi before a meeting at the State Pa
...
Subscribe to continue reading.
We craft news with stories.
 For the benefits of subscribing to Digital Tempo, See More
For the benefits of subscribing to Digital Tempo, See More








