Crying Over a False Result
Tuesday, May 12, 2020
arsip tempo : 173398527057.
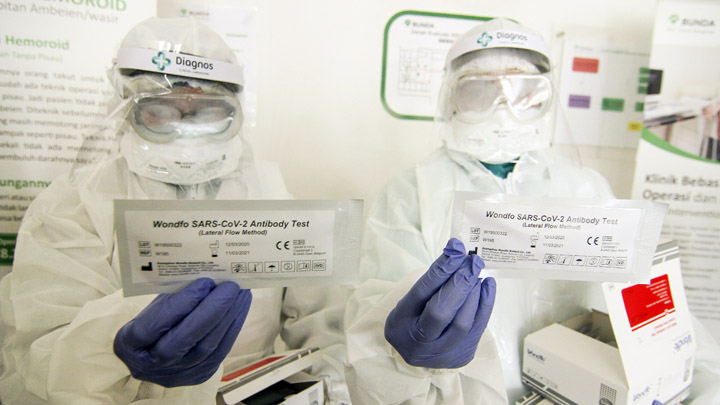
TIARA—not her real name—immediately wept when her rapid test gave a positive result for the coronavirus at the end of April. Only one week prior, another rapid test performed on the nurse at the Depok subdistrict community clinic (Puskesmas) in Sleman, Yogyakarta, yielded a negative result. “I instantly thought of my child, who is still a toddler,” she said on May 6.
The 36-year-old woman and her colleagues at the Depok P
...
Subscribe to continue reading.
We craft news with stories.
 For the benefits of subscribing to Digital Tempo, See More
For the benefits of subscribing to Digital Tempo, See More








