The Hunt for a Remedy
Tuesday, February 18, 2020
arsip tempo : 174542326178.
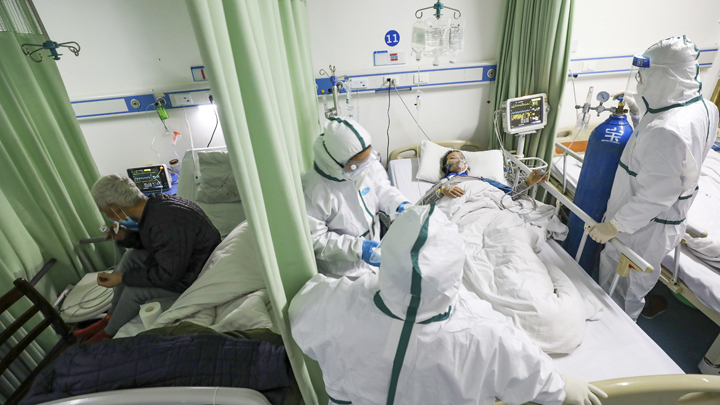
HUNDREDS of health experts from all over the world gathered at the World Health Organization (WHO) headquarters in Geneva, Switzerland, in mid-February. They discussed ways to address the Covid-19 virus, more popularly known as the coronavirus, which is currently plaguing China. “We must unite against this enemy,” said WHO Director-General Tedros Adhanom Ghebreyesus, as quoted on the WHO website on Wednesday, February 12.
Over 300 he
...
Subscribe to continue reading.
We craft news with stories.
 For the benefits of subscribing to Digital Tempo, See More
For the benefits of subscribing to Digital Tempo, See More








